[ad_1]
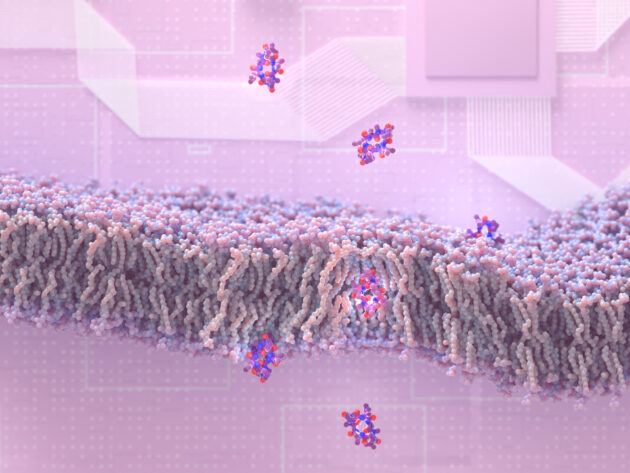
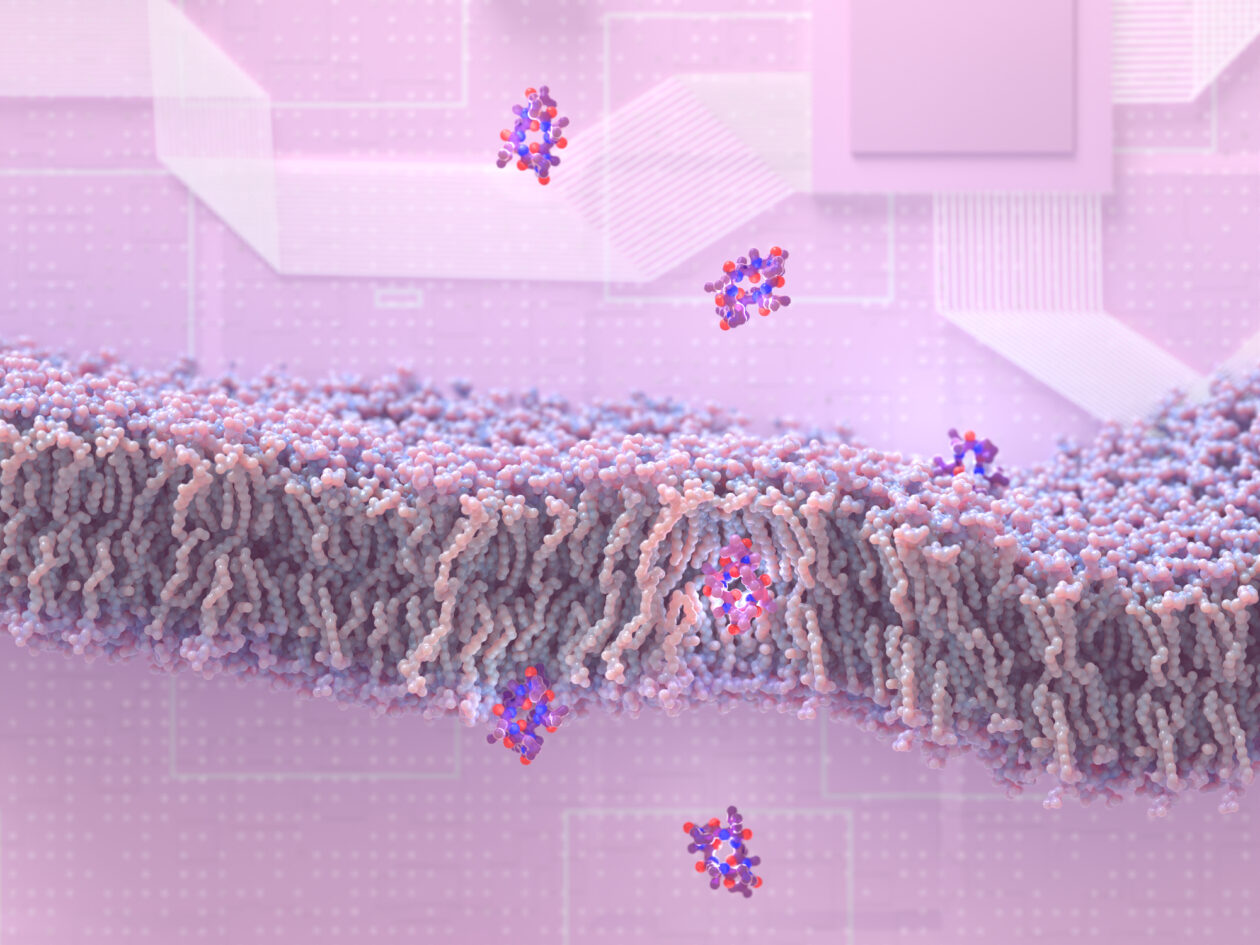
Researchers at the University of Washington have discovered how to create peptide molecules that can slip through membranes to enter cells — and they’ve also created a company with $50 million in backing to take advantage of the discovery for drug development.
The findings, which were published today in the journal Cell, could eventually lead to new types of oral medications for health disorders ranging from COVID-19 to cancer.
“This new ability to design membrane-permeable peptides with high structural accuracy opens the door to a new class of medicines that combine the advantages of traditional small-molecule drugs and larger protein therapeutics,” senior study author David Baker, a biochemist at the University of Washington School of Medicine, said in a news release.
The company, called Vilya, was formed by Baker and his research colleagues in collaboration with Arch Venture Partners. Vilya says it will license the platform and molecules described in the Cell research paper, and has raised $50 million in Series A financing from a group of investors led by Arch Venture Partners.
“Arch is thrilled to join forces with David to create a completely new class of medicines, one never before found in nature,” Robert Nelsen, a Vilya co-founder and Arch managing director, said in a news release. “It’s incredibly exciting to see real potential for this platform.”
Small-molecule drugs — for example, aspirin — are small enough to slip through cell membranes to do their work. Protein therapeutics — for example, monoclonal antibodies — can target more complex ailments, but the protein molecules are typically too big to wedge their way through lipid-based cell walls.
Peptide drugs are made from the same building blocks as protein, and offer many of the advantages of protein-based drugs. They can bind protein targets in the body more precisely than small-molecule drugs, promising fewer side effects.
“We know that peptides can be excellent medicines, but a big problem is that they don’t get into cells,” said study lead author Gaurav Bhardwaj, an assistant professor of medicinal chemistry at the UW School of Pharmacy. “There are a lot of great drug targets inside our cells, and if we can get in there, that space opens up.”
The newly reported experiments used a couple of molecular design techniques to create types of peptide molecules that can get into cells more easily.
Most peptides have chemical features that cause them to cling to water molecules instead of slipping through a cell’s lipid membrane. First, the researchers made synthetic peptides that were less likely to interact with water. They also designed peptides that could change shapes as they moved through membranes.
More than 180 custom-made peptide molecules were tested on artificial membranes in the lab. The researchers found that most of their peptides could pass through the lipids. Further lab tests, using gut epithelial cells, convinced the UW scientists that some of the molecules could make the jump from the stomach directly into the bloodstream.
Still more studies, conducted on mice and rats, showed that some of the peptides could efficiently move out of the gut, cross several membranes and enter living cells. Such peptides could theoretically be turned into oral medications. “These molecules are promising starting points for future drugs. My lab is now working to turn them into antibiotics, antivirals and cancer treatments,” Bhardwaj said.
Bhardwaj said peptide-based drugs could address the challenges posed by antibiotic resistance — and also offer a new strategy to fight COVID-19.
“One of the most obvious drug targets is located inside infected cells,” he said. “If we could shut down that enzyme, that would prevent the virus from creating more copies of itself.”
Bhardwaj and Baker are among several researchers at UW Medicine’s Institute for Protein Design who are part of the founding team for Vilya, along with several representatives from Arch Venture Partners. Steven Gillis, a managing director at Arch, is Vilya’s executive chair. (For what it’s worth, Vilya was the Elvish ring of power worn by Elrond in J.R.R. Tolkien’s “Lord of the Rings” saga.)
Vilya takes its place amid an array of companies created by researchers at the Institute for Protein Design — an array that also includes A-Alpha Bio, Arzeda, Cyrus Biotechnology, Icosavax, Lyell Immunopharma, Monod Bio, Mopad Biologics, Neoleukin Therapeutics, Outpace Bio (spun out from Lyell), PvP Biologics (acquired by Takeda Pharmaceuticals) and Sana Biotechnology.
Bhardwaj and Baker are among 26 authors of the paper published by Cell, titled “Accurate De Novo Design of Membrane-Traversing Macrocycles.”
The research was supported by The Audacious Project; Gates Ventures; Eric and Wendy Schmidt by recommendation of Schmidt Futures; the Nordstrom Barrier Institute for Protein Design Directors Fund; Wu Tsai Translational Fund; Bill and Melinda Gates Foundation; Takeda Pharmaceuticals; Howard Hughes Medical Institute; Washington State Supplement Funding; Department of Defense; Simons Foundation; Defense Threat Reduction Agency; National Institutes of Health; and Washington Research Foundation.
This report has been updated with a statement from Vilya.
[ad_2]
Source link